3D Medicines and ImmuneOncia Signed Exclusive License Agreement for Development, Manufacture and Commercialization of IMC-002 in Greater China
- 3D Medicines will lead the clinical development, manufacture and commercialization of IMC-002 for oncology in Greater China
- ImmuneOncia will receive potentially up to $470.5M payment including $8M upfront, and future development and commercial milestone payments, plus tiered royalties on net sales
Shanghai, March 31, 2021 - 3D Medicines Inc.(3DMed), a biopharmaceutical company focused on treating cancer as a chronic disease, and ImmuneOncia Therapeutics, Inc., a clinical-stage immuno-oncology company in South Korea, today announced the execution of an exclusive license agreement for the development, manufacture and commercialization of IMC-002, ImmuneOncia’s monoclonal antibody against CD47, for oncology indication in the Territory of Greater China (Mainland China, Hong Kong, Macau, and Taiwan). ImmuneOncia will retain rights to IMC-002 in the rest of the world including the United States, European Union, and Japan.

Under the terms of the agreement, ImmuneOncia will receive an upfront payment of $8million from 3DMed. ImmuneOncia is eligible to receive up to $462.5million upon the achievement of all future development and commercial milestones, plus tiered royalties up to double-digits on annual net sales of IMC-002 in Greater China. 3DMed is going to file the IND to NMPA this year.
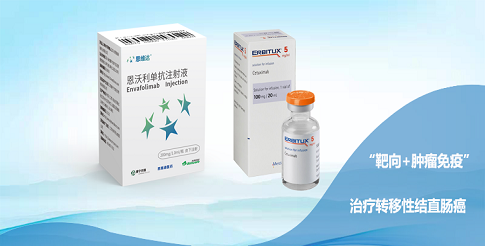
“We are pleased to enter into this exclusive collaboration with ImmuneOncia,” said John Gong, M.D., Ph.D., Chairman and Chief Executive Officer of 3DMed. “We believe that IMC-002, used in combination with existing standard of care therapeutics or Envafolimab, an innovative subcutaneous PD-L1 antibody which we have filed for marketing approval in China, could have certain clinical development potential. 3DMed is committed to exploring new treatment methods under the trend of treating cancer as a chronic disease, providing more treatment options for cancer patients, and helping people with cancer live longer and better.”
“3DMed is an established leader in research and development of oncology medicines, with a track record of successfully developing in-licensed oncology programs.” said Yun Jeong Song, Chief Executive Officer of ImmuneOncia. “We are confident that 3DMed is the ideal partner as we enter into our collaboration and we look forward to accelerating our delivery of IMC-002 to patients in Greater China.”
YAFO Capital (Shanghai) Co. Ltd. acted as financial advisor on this transaction for ImmuneOncia.

About IMC-002
IMC-002 is a fully human IgG4 monoclonal antibody designed to block the CD47–SIRPα interaction in order to promote the phagocytosis of cancer cells by macrophages. According to its non-clinical results, it binds to human CD47 with an optimal affinity that maximizes efficacy without binding to RBCs or causing anemia which is often seen in other CD47 blocking agents under development. For more information about the Phase 1 clinical trial, visit clinicaltrials.gov, identifier number NCT04306224.
About ImmuneOncia Therapeutics, Inc.
ImmuneOncia is an immuno-oncology-centric biopharmaceutical company. Established in 2016 as a joint venture company between Yuhan Corporation in South Korea and Sorrento Therapeutics, Inc. in U.S. ImmuneOncia leverages both companies’ expertise in drug development and antibody engineering. The company’s mission is to bring safe, effective, and novel immunotherapies to oncology patients world-wide, and its portfolio includes diverse immune checkpoint antibodies. ImmuneOncia has successfully completed a Phase I study of IMC-001, its leading candidate anti-PDL1 antibody, and a Phase II study was initiated in 2H 2020.
For more information, please visit www.immuneoncia.com.
About 3D Medicines Inc.
3D Medicines Inc. is a leading innovative biopharmaceutical company and committed to the development and commercialization of immuno-oncology (I/O) therapies with differentiated clinical benefit in response to the trend of treating cancer as a chronic disease. The mission is to help prolong the survival of cancer patients and improve their quality of life. The visionary management team has led us to build industry-leading, fully-integrated capabilities in end-to-end drug R&D from pre-clinical discovery to clinical development and commercialization. The core product envafolimab is a BLA-stage, subcutaneously-injectable PD-L1 antibody that has the potential to address a global unmet medical need for the treatment of cancer as a chronic disease. With a robust I/O monotherapy and combo-therapy portfolio of more than 10 drug candidates with envafolimab as the backbone, we strive to become a leader in the chronic cancer treatment market.
For more information, please visit www.3d-medicines.com.
Forward-looking Statement
The forward-looking statements made in this article relate only to the events or information as of the date on which the statements are made in this article. Except as required by law, we undertake no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this article completely and with the understanding that our actual future results or performance may be materially different from what we expect. In this article, statements of, or references to, our intentions or those of any of our Directors or our Company are made as of the date of this article. Any of these intentions may alter in light of future development.
热门文章

请咨询我们